Emerging Links Between Autism Spectrum Disorder and Alzheimer’s Disease:
- Fred Shaffer
- Apr 6
- 17 min read
When Development and Degeneration Converge

Appreciation
This post builds on two superb Washington Post articles by Ariana Eunjung Cha, "Surprising links between autism, Alzheimer’s could change how we treat both" and "New science points to 4 distinct types of autism."
Overview
Autism spectrum disorder (ASD) and Alzheimer’s disease (AD) have long been treated as conditions from different planets. One shapes how the brain is built in childhood; the other slowly dismantles it in old age. Clinicians trained in one field rarely cross into the other, and textbooks have kept them in separate chapters for decades.
That separation is starting to crack. A growing body of research reveals that these two conditions share far more biology than anyone anticipated, from overlapping genes to common breakdowns in the brain’s waste-disposal plumbing. The picture is still preliminary and, in some areas, speculative, but its practical implications are real and growing.
If the wall between neurodevelopment and neurodegeneration turns out to be artificial, then understanding how the brain is built may be essential to understanding how it falls apart. Interventions developed for one condition may illuminate treatments for the other. Healthcare practitioners who work with either population should pay attention, because these findings will increasingly shape screening, monitoring, and long-term care.
Epidemiological Evidence: Higher Dementia Risk in Autism Spectrum Disorder Adults
Autism was formally recognized as a diagnosis in 1980, and for decades, it was treated almost exclusively as a childhood condition. Researchers are only now beginning to ask what happens to autistic adults as they age, and the answer is unsettling.
Of the more than 40,000 autism papers published between 1980 and 2021, only a tiny fraction included participants over 50 (Cha, 2026). That gap has left clinicians without evidence-based guidance for a population that may number nearly two million adults in the United States alone (Centers for Disease Control and Prevention, 2024).
The first large-scale study to look at this question examined more than 1.2 million adults on Medicaid between ages 30 and 64. Vivanti et al. (2021) found that adults with ASD were roughly 2.6 times more likely to be diagnosed with early-onset dementia than adults without ASD, even after accounting for depression, cardiovascular risk, and other known factors. Those with both ASD and intellectual disability faced nearly triple the risk.
Critically, the elevated risk was not explained away by lifestyle, psychiatric history, or other medical conditions. Something about autism itself appears to confer additional vulnerability to cognitive decline.
The age at which dementia was diagnosed was also striking. Autistic adults received their dementia diagnosis at an average age of about 49, compared to nearly 54 in the general Medicaid population (Vivanti et al., 2021). These results were replicated in 2025 using combined Medicaid and Medicare data (Cha, 2026).
Lifestyle factors undoubtedly play a role. Barriers to exercise, social isolation, communication difficulties that complicate medical care, and higher lifetime stress all deserve consideration (Cha, 2026). But these explanations alone do not account for the full pattern. The biology itself appears to be involved, and the evidence for that involvement keeps accumulating.
Shared Genes: A Growing List of Molecular Overlaps
When researchers began comparing the genetic underpinnings of autism and Alzheimer’s, the degree of overlap was startling. A 2025 analysis found that the two conditions share at least 148 genes, many of which are tied to fundamental processes that build and sustain the brain over a lifetime (Trifonova et al., 2025).
These are not obscure genes on the margins of brain function. They sit at the heart of how neurons connect, communicate, and clean up after themselves.
One especially revealing example is SHANK3, a gene that encodes a scaffolding protein essential for healthy synapses. In autism, mutations in SHANK3 disrupt synaptic connections during early development. In Alzheimer’s, SHANK3 protein levels decline as the disease progresses, and that decline tracks with the gradual loss of neural connections (Cha, 2026). The same molecular machinery that builds synapses early in life is also needed to keep them running later on.

Even more striking is what happens when researchers zoom out from individual genes to the pathways those genes feed into. Nearly half of autism predisposition genes and about two-fifths of Alzheimer’s genes connect to a single master regulatory system called mTOR, which controls cell growth, protein production, and the process by which cells clear out their own debris (Trifonova et al., 2025).

This finding led the authors to propose that both conditions may be “mTORopathies.” The label matters because drugs that target mTOR already exist and have been tested in related conditions.
Evolutionary analysis adds a further twist. The genes disrupted in autism are not newcomers. They are among the most ancient and conserved genes in the animal kingdom, preserved across hundreds of millions of years of evolution (Trifonova et al., 2025). Their conservation signals that the pathways they regulate are not peripheral features of the nervous system but foundational ones.
Family studies reinforce the genetic connection. Chang et al. (2025) found that families with members who have Alzheimer’s are more likely to have children with autism. Lee and Seong (2024) used a statistical technique called Mendelian randomization to identify synaptic dysfunction as a shared genetic mechanism linking the two conditions.
Synaptic Connections: Where Development and Degeneration Meet
Zoom in far enough on both conditions and you arrive at the same place: the synapse, the microscopic gap where one neuron hands a signal to the next. In autism, synaptic connections form differently during development. In Alzheimer’s, those same connections gradually come undone. The surprise is that many of the same molecular players are involved in both processes.
Research presented in 2025 by James McPartland of Yale found that adults with autism had fewer synapses than their neurotypical peers, and that those with the fewest connections struggled most with the social demands of daily life (Cha, 2025). This mirrors Alzheimer’s research, where synaptic loss now appears to be a better predictor of cognitive decline than the amyloid plaques and tau tangles that have long defined the disease.
At Mount Sinai, Buxbaum’s lab has been studying mice with the SHANK3 mutations associated with autism. As these mice age, they develop cognitive inflexibility, an early hallmark of Alzheimer’s. Yet paradoxically, they resist full-blown dementia-like pathology (Cha, 2026). If researchers can figure out what protects these brains despite their synaptic differences, they may find clues that could eventually help humans with either condition.
Meanwhile, at the Gladstone Institutes in San Francisco, researchers found that cutting tau protein levels in half prevented core autism symptoms in mouse models of severe ASD, and the effect lasted a lifetime (Cha, 2026).

Tau has traditionally been Alzheimer’s territory, but these findings place it squarely in the developmental story of autism as well. The proteins that build synapses during development and the proteins that maintain them in aging turn out to be the same cast of characters.
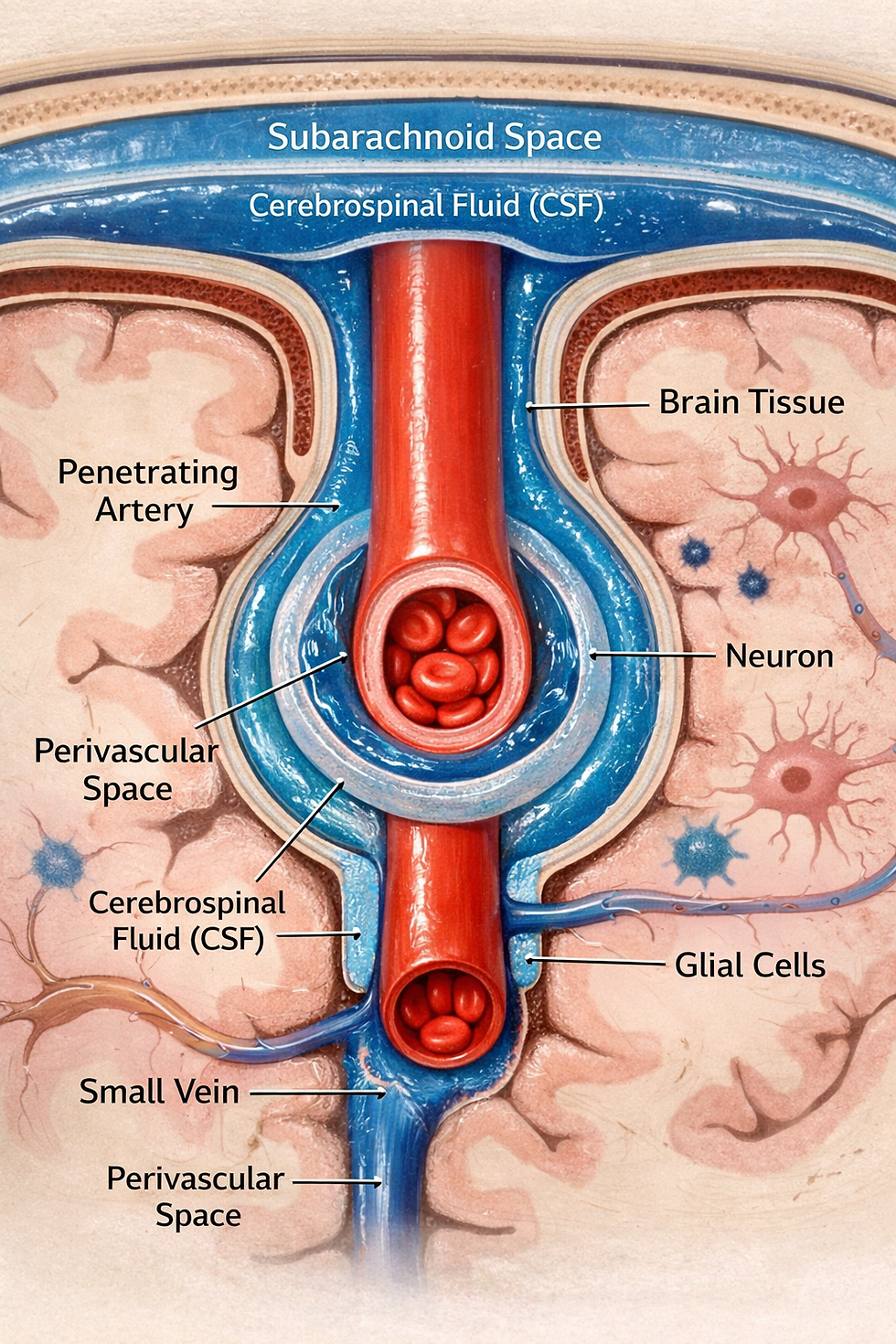
The Brain’s Waste-Clearance System: Glymphatic Dysfunction in Both Conditions
Every night while you sleep, your brain runs its own cleaning cycle. A network of channels surrounding the brain’s blood vessels flushes cerebrospinal fluid through brain tissue, carrying away toxic proteins like amyloid-beta and tau. This system, called the glymphatic system, was only discovered in 2012 (Iliff et al., 2012). When it malfunctions, waste builds up, proteins misfold, and neural communication degrades.

Phillips et al. (2026) published a paper documenting extensive parallels in glymphatic dysfunction between autism and Alzheimer’s. Both conditions show enlarged, fluid-filled channels around blood vessels and excess cerebrospinal fluid pooling between the brain and the skull, signs that the brain’s drainage system is not functioning properly (Garic et al., 2023; Menze et al., 2024; Shen, 2018).
Specialized MRI techniques confirm the pattern: both autistic children and Alzheimer’s patients show reduced efficiency of glymphatic clearance compared to age-matched controls (Huang et al., 2024; Li et al., 2022; Zhao et al., 2025).
The timing of these abnormalities in autism is remarkable. Excess fluid between the brain and skull is detectable as early as 6 months of age in infants who later develop autism, long before any behavioral signs appear (Shen et al., 2013, 2017).
By 24 months, enlarged perivascular spaces are associated with more frequent night wakings in school-age children (Garic et al., 2023). This connects the brain’s plumbing problems to the sleep disturbances that are common in both ASD and AD.
Sleep is central to this story because the glymphatic system is most active during sleep (Xie et al., 2013). Both the autistic and Alzheimer’s populations have high rates of sleep disruption. They also share other risk factors for glymphatic impairment, including high blood pressure, elevated body mass index, and altered heart rate variability (Bharath et al., 2019; Croen et al., 2015; Thapa et al., 2021).
A key molecular player in glymphatic clearance is aquaporin-4 (AQP4), the brain’s primary water channel. Altered AQP4 expression has been documented in both autistic brains and in Alzheimer’s (Beer et al., 2024; Fatemi et al., 2008). In rats, chronic blockade of AQP4 produces autism-like behaviors (Davoudi et al., 2023). Phillips et al. (2026) further propose that swelling of blood vessels in the nasal passages may obstruct a major drainage route for cerebrospinal fluid, though this nasal obstruction hypothesis remains speculative, particularly in ASD.
Neuroimaging Patterns: Convergent Brain Architectures
For years, brain imaging research in autism and Alzheimer’s focused on which regions were bigger or smaller, more or less active. Some findings seemed to point in opposite directions. Alzheimer’s shrinks the amygdala; in autism, the amygdala is often enlarged. Young children with autism sometimes show unusually rapid brain growth in infancy (Cha, 2025, 2026).
But as imaging technology has matured, the focus has shifted from individual regions to the connections between them. In both fields, researchers have independently arrived at the same conclusion: it is the pattern of connectivity, not the size of any single structure, that best predicts how people actually function (Cha, 2026). That convergence was invisible as long as the two research communities stayed in their own lanes.
B. Blair Braden at Arizona State University has spent more than a decade scanning the brains of autistic adults over time. Her work revealed accelerated shrinkage of the hippocampus, the brain’s episodic memory hub and one of the earliest casualties of Alzheimer’s (Cha, 2026). The finding suggests that some autistic brains may age more rapidly in regions critical to cognitive health.
Both conditions also show decreased glucose metabolism in the brain, a sign that neurons are not getting the fuel they need (Keeratitanont et al., 2022; Mosconi, 2005). In Alzheimer’s disease, this metabolic slowdown can appear years before clinical symptoms emerge (Mosconi et al., 2008). Both also show olfactory dysfunction, which has traditionally been interpreted differently in each field but may reflect the same underlying glymphatic drainage problem (Bennetto et al., 2007; Fatuzzo et al., 2023; Phillips et al., 2026).
Therapeutic Implications: Learning from One Condition to Treat the Other
The shared biology between autism and Alzheimer’s is not just an intellectual curiosity. It is already pointing researchers toward treatments that might never have been considered if the two fields had stayed in their separate silos. PubMed contains more than 86,000 publications on Alzheimer’s treatment but fewer than 27,000 on autism treatment (Trifonova et al., 2025). If the shared molecular pathways hold up, Alzheimer’s research could become a rich source of therapeutic leads for autism.
The mTOR pathway is the most developed therapeutic avenue. Rapamycin, a drug that inhibits mTOR, reversed behavioral and learning deficits in a common mouse model of syndromic autism (Ehninger et al., 2008) and reduced amyloid plaques and tangles in Alzheimer’s mice (Caccamo et al., 2010). The anti-inflammatory drug celecoxib, another mTOR modulator, has already shown benefit for autism symptoms in a randomized controlled trial (Asadabadi et al., 2013). Trifonova et al. (2025) identified a wider list of mTOR-targeting compounds, both pharmaceutical and natural, that deserve further study.
Tau-targeted therapies offer another bridge between the two conditions. If cutting tau levels in half can prevent autism symptoms in mice for a lifetime, as the Gladstone Institutes showed (Cha, 2026), then tau-lowering drugs being developed for Alzheimer’s may also benefit autistic individuals. Meanwhile, glymphatic-targeted therapies are emerging from an entirely different direction. Noninvasive massage of cervical lymphatics significantly increased brain waste clearance in one study (Jin et al., 2025), and topical prostaglandin application restored lymphatic flow in aged mice (Du et al., 2024).
For clinicians, the practical takeaway is straightforward. Healthcare practitioners working with autistic adults should watch for signs of cognitive decline, especially after age 40 in individuals with co-occurring intellectual disability, depression, or cardiovascular risk factors (Vivanti et al., 2021). Sleep quality assessment deserves special attention, given the glymphatic system’s dependence on sleep (Xie et al., 2013).
A neurodiversity-affirming approach to care does not preclude vigilance about neurological health across the lifespan.
Conclusion
The emerging connections between autism and Alzheimer’s challenge one of neuroscience’s oldest assumptions: that how the brain develops and how it degenerates are separate stories. The evidence now spans epidemiology, genetics, synaptic biology, brain waste clearance, neuroimaging, and therapeutic response. For healthcare practitioners, these findings carry concrete implications for how we screen, monitor, and treat both populations across the lifespan.
Key Takeaways
1. Adults with autism are approximately 2.6 times more likely to develop early-onset dementia than the general population, even after adjusting for known risk factors.
2. At least 148 genes are shared between ASD and AD, with nearly half of autism predisposition genes and two-fifths of Alzheimer’s genes linked to the mTOR signaling pathway, making both conditions potential candidates for mTOR-targeted therapies.
3. The brain’s glymphatic waste-clearance system is impaired in both conditions, detectable through MRI, and may represent a shared mechanism that could be targeted therapeutically.
4. Synaptic density and brain connectivity patterns, rather than the size of individual brain regions, may provide the most meaningful framework for understanding and tracking both conditions.
5. Clinicians serving autistic adults should incorporate age-related cognitive monitoring into routine care, with particular attention to sleep quality, cardiovascular health, and early signs of cognitive decline beginning in the fourth decade of life.
Glossary
amygdala: an almond-shaped brain structure involved in emotion processing, detecting salient stimuli, fear responses, and social behavior.
amyloid-beta (Aβ): a protein fragment that accumulates into plaques in the brains of individuals with Alzheimer’s disease, disrupting neuronal communication.
aquaporin-4 (AQP4): the principal water channel protein in the central nervous system, located on astrocytic endfeet surrounding blood vessels, that facilitates water transport and glymphatic clearance.
cerebrospinal fluid (CSF): a clear fluid that surrounds and cushions the brain and spinal cord, transporting nutrients and removing waste products.
enlarged perivascular spaces (EPVS): fluid-filled channels surrounding blood vessels in the brain that become visibly enlarged on MRI when glymphatic clearance is impaired.
glymphatic system: a brain-wide network of perivascular channels that facilitates the clearance of metabolic waste from the brain, primarily active during sleep.
hippocampus: a brain structure critical for memory formation and spatial navigation, among the earliest regions affected in Alzheimer’s disease.
Mendelian randomization: a statistical method that uses genetic variants as natural experiments to determine whether an observed association between a risk factor and a disease is causal.
mTOR (mechanistic target of rapamycin): a master regulatory protein kinase that controls cell growth, protein synthesis, autophagy, and metabolism. Dysregulation is implicated in both ASD and AD.
perivascular spaces: fluid-filled channels that surround blood vessels as they penetrate the brain, serving as conduits for glymphatic waste clearance.
SHANK3: a gene encoding a scaffolding protein critical for synaptic structure and function, implicated in both autism (through developmental mutations) and Alzheimer’s disease (through age-related protein decline).
synapse: the junction between two neurons where electrical or chemical signals are transmitted, enabling neural communication.
tau: a protein that stabilizes microtubules within neurons. In Alzheimer’s disease, tau becomes hyperphosphorylated and forms neurofibrillary tangles. Tau pathology has also been identified in some forms of autism.
References
Asadabadi, M., Mohammadi, M. R., Ghanizadeh, A., Modabbernia, A., Ashrafi, M., Hassanzadeh, E., Forghani, S., & Akhondzadeh, S. (2013). Celecoxib as adjunctive treatment to risperidone in children with autistic disorder: A randomized, double-blind, placebo-controlled trial. Psychopharmacology, 225(1), 51–59. https://doi.org/10.1007/s00213-012-2796-8
Beer, S., Elmenhorst, D., Bischof, G. N., Ramirez, A., Bauer, A., Drzezga, A., & Düzel, E. (2024). Explainable artificial intelligence identifies an AQP4 polymorphism-based risk score associated with brain amyloid burden. Neurobiology of Aging, 143, 19–29. https://doi.org/10.1016/j.neurobiolaging.2024.08.002
Bennetto, L., Kuschner, E. S., & Hyman, S. L. (2007). Olfaction and taste processing in autism. Biological Psychiatry, 62(9), 1015–1021. https://doi.org/10.1016/j.biopsych.2007.04.019
Bharath, R., Moodithaya, S. S., Bhat, S. U., Mirajkar, A. M., & Shetty, S. B. (2019). Comparison of physiological and biochemical autonomic indices in children with and without autism spectrum disorders. Medicina, 55(7), 346. https://doi.org/10.3390/medicina55070346
Caccamo, A., Majumder, S., Richardson, A., Strong, R., & Oddo, S. (2010). Molecular interplay between mammalian target of rapamycin (mTOR), amyloid-beta, and tau: Effects on cognitive impairments. Journal of Biological Chemistry, 285(17), 13107–13120. https://doi.org/10.1074/jbc.M110.100420
Centers for Disease Control and Prevention. (2024). Autism data and statistics. https://www.cdc.gov/autism/data-research/
Cha, A. E. (2025, December 26). New science points to 4 distinct types of autism. The Washington Post.
Cha, A. E. (2026, April 6). Surprising links between autism, Alzheimer’s could change how we treat both. The Washington Post.
Chang, Z., Yao, H., Sun, S., Zhang, L., Liu, S., Brikell, I., Almqvist, C., & Larsson, H. (2025). Association between autism and dementia across generations: Evidence from a family study of the Swedish population. Molecular Psychiatry, 30, 4605–4612. https://doi.org/10.1038/s41380-025-03045-6
Cortese, S., Wang, F., Angriman, M., Masi, G., & Bruni, O. (2020). Sleep disorders in children and adolescents with autism spectrum disorder: Diagnosis, epidemiology, and management. CNS Drugs, 34(4), 415–423. https://doi.org/10.1007/s40263-020-00710-y
Croen, L. A., Zerbo, O., Qian, Y., Massolo, M. L., Rich, S., Sidney, S., & Kripke, C. (2015). The health status of adults on the autism spectrum. Autism, 19(7), 814–823. https://doi.org/10.1177/1362361315577517
Crow, A. J. D., Janssen, J. M., Vickers, K. L., Parish-Morris, J., Moberg, P. J., & Roalf, D. R. (2020). Olfactory dysfunction in neurodevelopmental disorders: A meta-analytic review of autism spectrum disorders, attention deficit/hyperactivity disorder and obsessive-compulsive disorder. Journal of Autism and Developmental Disorders, 50(8), 2685–2697. https://doi.org/10.1007/s10803-020-04376-9
Cunnane, S., Nugent, S., Roy, M., Courchesne-Loyer, A., Croteau, E., Tremblay, S., Castellano, A., Pifferi, F., Bocti, C., Paquet, N., Begdouri, H., Bentourkia, M., Turcotte, E., Allard, M., Barberger-Gateau, P., Fulop, T., & Rapoport, S. I. (2011). Brain fuel metabolism, aging, and Alzheimer’s disease. Nutrition, 27(1), 3–20. https://doi.org/10.1016/j.nut.2010.07.021
Davoudi, S., Rahdar, M., Hosseinmardi, N., Behzadi, G., & Janahmadi, M. (2023). Chronic inhibition of astrocytic aquaporin-4 induces autistic-like behavior in control rat offspring similar to maternal exposure to valproic acid. Physiology & Behavior, 269, 114286. https://doi.org/10.1016/j.physbeh.2023.114286
Du, T., Raghunandan, A., Mestre, H., Plá, V., Liu, G., Ladrón-de-Guevara, A., Newbold, E., Tobin, P., Gahn-Martinez, D., Pattanayak, S., Huang, Q., Peng, W., Nedergaard, M., & Kelley, D. H. (2024). Restoration of cervical lymphatic vessel function in aging rescues cerebrospinal fluid drainage. Nature Aging, 4, 1418–1431. https://doi.org/10.1038/s43587-024-00691-3
Ehninger, D., Han, S., Shilyansky, C., Zhou, Y., Li, W., Kwiatkowski, D. J., Ramesh, V., & Silva, A. J. (2008). Reversal of learning deficits in a Tsc2+/− mouse model of tuberous sclerosis. Nature Medicine, 14(8), 843–848. https://doi.org/10.1038/nm1788
Fatemi, S. H., Folsom, T. D., Reutiman, T. J., & Lee, S. (2008). Expression of astrocytic markers aquaporin 4 and connexin 43 is altered in brains of subjects with autism. Synapse, 62(7), 501–507. https://doi.org/10.1002/syn.20519
Fatuzzo, I., Niccolini, G. F., Zoccali, F., Cavalcanti, L., Bellizzi, M. G., Riccardi, G., & De Vincentiis, M. (2023). Neurons, nose, and neurodegenerative diseases: Olfactory function and cognitive impairment. International Journal of Molecular Sciences, 24(3), 2117. https://doi.org/10.3390/ijms24032117
Garic, D., McKinstry, R. C., Rutsohn, J., Slomowitz, R., Wolff, J., MacIntyre, L. C., Steinman, K. J., King, T. Z., Dager, S. R., Estes, A. M., Gerig, G., Hazlett, H. C., McKinstry, R. C., Pandey, J., Schultz, R. T., Styner, M., Zwaigenbaum, L., Piven, J., Shen, M. D., & the IBIS Network. (2023). Enlarged perivascular spaces in infancy and autism diagnosis, cerebrospinal fluid volume, and later sleep problems. JAMA Network Open, 6(12), e2348341. https://doi.org/10.1001/jamanetworkopen.2023.48341
Grigg, I., Ivashko-Pachima, Y., Hait, T. A., Korenková, V., Touloumi, O., Lagoudaki, R., Van Dijck, A., Marusic, Z., Anicic, M., Vukovic, J., Kooy, R. F., Grigoriadis, N., & Gozes, I. (2020). Tauopathy in the young autistic brain: Novel biomarker and therapeutic target. Translational Psychiatry, 10, 228. https://doi.org/10.1038/s41398-020-00904-4
Helsmoortel, C., Vulto-van Silfhout, A. T., Coe, B. P., Vandeweyer, G., Rooms, L., van den Ende, J., & Eichler, E. E. (2014). A SWI/SNF-related autism syndrome caused by de novo mutations in ADNP. Nature Genetics, 46(4), 380–384. https://doi.org/10.1038/ng.2899
Huang, S. Y., Ge, Y. J., Ren, P., Wu, B. S., Gong, W., Du, J., & Yu, J. T. (2025). Genome-wide association study unravels mechanisms of brain glymphatic activity. Nature Communications, 16, 626. https://doi.org/10.1038/s41467-024-55706-9
Huang, S. Y., Zhang, Y. R., Guo, Y., Du, J., Ren, P., Wu, B. S., & Yu, J. T. (2024). Glymphatic system dysfunction predicts amyloid deposition, neurodegeneration, and clinical progression in Alzheimer’s disease. Alzheimer’s & Dementia, 20(5), 3251–3269. https://doi.org/10.1002/alz.13789
Iliff, J. J., Wang, M., Liao, Y., Plogg, B. A., Peng, W., Gundersen, G. A., Benveniste, H., Vates, G. E., Deane, R., Goldman, S. A., Nagelhus, E. A., & Nedergaard, M. (2012). A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Science Translational Medicine, 4(147), 147ra111. https://doi.org/10.1126/scitranslmed.3003748
Jessup, M., Iglesias, N. J., Tadisina, K. K., Xu, K., Mella-Catinchi, J., Pinchuk, L., Singh, D., Tse, D. T., Nayak, V. V., & Coelho, P. G. (2025). The emerging role of biomaterial applications in cerebral lymphatic surgical interventions: A narrative review. Biomaterials and Biosystems, 19, 100117. https://doi.org/10.1016/j.bbiosy.2025.100117
Jin, H., Yoon, J. H., Hong, S. P., Hwang, Y. S., Yang, M. J., Choi, J., & Koh, G. Y. (2025). Increased CSF drainage by non-invasive manipulation of cervical lymphatics. Nature, 643, 755–767. https://doi.org/10.1038/s41586-025-09052-5
Johnston, M., Zakharov, A., Papaiconomou, C., Salmasi, G., & Armstrong, D. (2004). Evidence of connections between cerebrospinal fluid and nasal lymphatic vessels in humans, non-human primates and other mammalian species. Cerebrospinal Fluid Research, 1, 2. https://doi.org/10.1186/1743-8454-1-2
Keeratitanont, K., Theerakulpisut, D., Auvichayapat, N., Suphakunpinyo, C., Patjanasoontorn, N., Tiamkao, S., & Auvichayapat, P. (2022). Brain laterality evaluated by F-18 fluorodeoxyglucose positron emission computed tomography in autism spectrum disorders. Frontiers in Molecular Neuroscience, 15, 901016. https://doi.org/10.3389/fnmol.2022.901016
Lee, J., & Seong, I. S. (2024). Mendelian randomization reveals shared genetic landscape in autism spectrum disorder and Alzheimer’s disease. Journal of Emerging Investigators. https://doi.org/10.59720/23-248
Li, X., Ruan, C., Zibrila, A. I., Musa, M., Wu, Y., Zhang, Z., & Zhang, Z. (2022). Children with autism spectrum disorder present glymphatic system dysfunction evidenced by diffusion tensor imaging along the perivascular space. Medicine, 101(49), e32061. https://doi.org/10.1097/MD.0000000000032061
Menze, I., Bernal, J., Kaya, P., Aki, C., Pfister, M., Geisendorfer, J., & Wirth, M. (2024). Perivascular space enlargement accelerates in ageing and Alzheimer’s disease pathology: Evidence from a three-year longitudinal multicentre study. Alzheimer’s Research & Therapy, 16, 242. https://doi.org/10.1186/s13195-024-01603-8
Mosconi, L. (2005). Brain glucose metabolism in the early and specific diagnosis of Alzheimer’s disease. European Journal of Nuclear Medicine and Molecular Imaging, 32(4), 486–510. https://doi.org/10.1007/s00259-005-1762-7
Mosconi, L., Pupi, A., & De Leon, M. J. (2008). Brain glucose hypometabolism and oxidative stress in preclinical Alzheimer’s disease. Annals of the New York Academy of Sciences, 1147, 180–195. https://doi.org/10.1196/annals.1427.007
Nadeem, M. S., Hosawi, S., Alshehri, S., Ghoneim, M. M., Imam, S. S., Murtaza, B. N., & Kazmi, I. (2021). Symptomatic, genetic, and mechanistic overlaps between autism and Alzheimer’s disease. Biomolecules, 11(11), 1635. https://doi.org/10.3390/biom11111635
Nagra, G., Koh, L., Zakharov, A., Armstrong, D., & Johnston, M. (2006). Quantification of cerebrospinal fluid transport across the cribriform plate into lymphatics in rats. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 291(5), R1383–R1389. https://doi.org/10.1152/ajpregu.00235.2006
Phillips, W. T., Issa, N. J., Elhalwagi, S. B., Draeger, H. T., Schwartz, J. G., & Gelfond, J. A. (2022). Nasal and parotid blood pool activity is significantly correlated with metabolic syndrome components and sleep apnea. Metabolic Syndrome and Related Disorders, 20(7), 395–404. https://doi.org/10.1089/met.2022.0015
Phillips, W. T., Sheesley, A. P., & Schwartz, J. G. (2026). Pathophysiologic similarities between autism spectrum disorder and Alzheimer’s disease: Therapeutic possibilities. Frontiers in Neuroscience, 19, 1737007. https://doi.org/10.3389/fnins.2025.1737007
Plog, B. A., Kim, K., Verhaege, D., Kim, M. W., Papadopoulos, Z., Dikranian, K., Dykstra, T., Cao, J., Perrin, R. J., Schwetye, K. E., Kipnis, J., & Drieu, A. (2025). A route for cerebrospinal fluid flow through leptomeningeal arterial-venous overlaps enables macromolecule and fluid shunting. Nature Neuroscience, 28, 1436–1445. https://doi.org/10.1038/s41593-025-01977-4
Reynolds, A. M., & Malow, B. A. (2011). Sleep and autism spectrum disorders. Pediatric Clinics of North America, 58(3), 685–698. https://doi.org/10.1016/j.pcl.2011.03.009
Shen, M. D. (2018). Cerebrospinal fluid and the early brain development of autism. Journal of Neurodevelopmental Disorders, 10, 39. https://doi.org/10.1186/s11689-018-9256-7
Shen, M. D., Kim, S. H., McKinstry, R. C., Gu, H., Hazlett, H. C., Nordahl, C. W., & Piven, J. (2017). Increased extra-axial cerebrospinal fluid in high-risk infants who later develop autism. Biological Psychiatry, 82(3), 186–193. https://doi.org/10.1016/j.biopsych.2017.02.1095
Shen, M. D., Nordahl, C. W., Young, G. S., Wootton-Gorges, S. L., Lee, A., Liston, S. E., & Amaral, D. G. (2013). Early brain enlargement and elevated extra-axial fluid in infants who develop autism spectrum disorder. Brain, 136(9), 2825–2835. https://doi.org/10.1093/brain/awt166
Thapa, R., Pokorski, I., Ambarchi, Z., Thomas, E., Demayo, M., Boulton, K., & Guastella, A. J. (2021). Heart rate variability in children with autism spectrum disorder and associations with medication and symptom severity. Autism Research, 14(1), 75–85. https://doi.org/10.1002/aur.2437
Trifonova, E. A., Pashchenko, A. A., Ivanov, R. A., Kochetov, A. V., & Lashin, S. A. (2025). Genetic and pathogenic overlaps between autism spectrum disorder and Alzheimer’s disease: Evolutionary features and opportunities for drug repurposing. International Journal of Molecular Sciences, 26(20), 10066. https://doi.org/10.3390/ijms262010066
Vivanti, G., Tao, S., Lyall, K., Robins, D. L., & Shea, L. L. (2021). The prevalence and incidence of early-onset dementia among adults with autism spectrum disorder. Autism Research, 14(10), 2189–2199. https://doi.org/10.1002/aur.2590
Xie, L., Kang, H., Xu, Q., Chen, M. J., Liao, Y., Thiyagarajan, M., O’Donnell, J., Christensen, D. J., Nicholson, C., Iliff, J. J., Takano, T., Deane, R., & Nedergaard, M. (2013). Sleep drives metabolite clearance from the adult brain. Science, 342(6156), 373–377. https://doi.org/10.1126/science.1241224
Zhao, S., Zhang, M., Luo, T., Li, L., Jiang, Y., Situ, M., & Huang, Y. (2025). Glymphatic system dysfunction in children with autism spectrum disorder as evidenced by the diffusion tensor imaging along perivascular spaces index. Frontiers in Psychiatry, 16, 1701816. https://doi.org/10.3389/fpsyt.2025.1701816
About the Author
Fred Shaffer earned his PhD in Psychology from Oklahoma State University. He earned BCIA certifications in Biofeedback and HRV Biofeedback. Fred is an Allen Fellow and Professor of Psychology at Truman State University, where he has taught for 50 years. He is a Biological Psychologist who consults and lectures in heart rate variability biofeedback, Physiological Psychology, and Psychopharmacology. Fred helped to edit Evidence-Based Practice in Biofeedback and Neurofeedback (3rd and 4th eds.) and helps to maintain BCIA's certification programs. He is a recipient of AAPB's Distinguished Scientist Award and BFE's Lifetime Impact Award.

Support Our Friends







Comments