Continuous Glucose Monitoring: Biofeedback for Type 2 Diabetes Patients
- Fred Shaffer
- 1 hour ago
- 20 min read

Executive Summary
A small wire under the skin is rewriting how clinicians and patients understand Type 2 diabetes. Continuous glucose monitoring (CGM) provides glycemic biofeedback to patients with Type 2 diabetes and to healthy individuals who want to optimize their health and performance.
Biofeedback teaches clients to tune into their bodies. Over time, they rely less on external displays and more on internal sensations.

CGM once lived in specialty endocrinology clinics, paired with insulin pumps and prescribed cautiously. It has now walked into pharmacies and onto wrists, propelled by FDA clearance of the first over-the-counter sensor and by professional standards that encourage broader use across the diabetes care continuum (American Diabetes Association Professional Practice Committee for Diabetes, 2026b; U.S. Food and Drug Administration, 2024b).
For adults with Type 2 diabetes, the case for continuous monitoring is strongest when it surfaces daily glucose patterns that finger-stick testing and laboratory averages routinely miss (Jancev et al., 2024; Martens et al., 2021; Uhl et al., 2024).
The science behind avoiding repeated post-meal spikes is grounded in vascular biology and supported by guideline targets for time in range (Battelino et al., 2019; International Diabetes Federation, 2011; Raj et al., 2022). Yet 160 mg/dL is best understood as a conservative early-warning line, not a magic injury threshold.

Many consensus reports define clinically important high readings as time spent above 180 mg/dL. The distinction matters in clinical conversation. A single brief peak is not proof of harm; a repeated pattern of peaks deserves attention.
Continuous monitoring can reveal silent excursions between 160 and 180 mg/dL that patients never feel and that quarterly hemoglobin A1c values hide. That visibility becomes valuable when it leads to specific, testable experiments with food order, walking, sleep, and medication review (Engeroff et al., 2023; Shukla et al., 2018; Touhamy et al., 2025). The same visibility can become harmful when it generates uninterpreted alarms, perfectionism, or shame (Ehrmann et al., 2024; Spartano et al., 2025; Yue, 2026).
Clients should not chase every glucose peak, but rather turn patterns into safe, psychologically informed decisions. Behavioral health clinicians have a particular contribution to make. They can help clients receive sensor data as feedback rather than surveillance, especially when shame, perfectionism, disordered eating, or diabetes distress are already in the room (Zu et al., 2024).
A Sensor Turns Glucose Into a Story
Continuous glucose monitoring (CGM) is a wearable sensing system that estimates glucose levels around the clock through a small filament inserted just under the skin.

In Type 2 diabetes, a chronic metabolic condition in which insulin resistance and declining insulin production drive glucose levels upward, this technology turns an invisible bodily signal into a readable pattern.

Monitoring your glucose levels in real time is glycemic biofeedback, meaning real-time information that a patient uses to adjust meals, movement, sleep, and conversations with the prescribing clinician.

The sensor does not measure blood directly. Instead, it samples glucose in interstitial fluid, the watery medium that surrounds and bathes body cells. Glucose travels from the blood to the interstitial fluid with a brief delay, resulting in sensor lag. During steady periods, the lag is barely noticeable. During rapid changes, such as in the minutes after drinking juice or exercising, the screen value can lag behind the true value by 10 to 15 minutes. Clinicians who teach this concept up front spare patients the frustration of having to distrust their device.

This distinction shapes how a CGM reading should be interpreted.
The number on the screen is not a moral score, a diagnosis, or a perfect laboratory value. It is a trend line. The most useful clinical question is rarely "What is the value?" It is closer to "What pattern is this showing, and what safe experiment could we run together?"
Two forces have driven the rapid uptake of CGM. People with diabetes want richer feedback than a single morning finger stick can provide. People without diabetes are increasingly curious about glucose as a wellness metric. FDA clearance of the first over-the-counter sensor expanded access for the second group, while updated diabetes technology standards encourage earlier and broader consideration of CGM in clinical care (American Diabetes Association Professional Practice Committee for Diabetes, 2026b; U.S. Food and Drug Administration, 2024b; Yue, 2026).
Healthy users may discover that soda, a late dinner, poor sleep, or a stressful meeting nudges their glucose curve. They may not know what to do with that observation. There is still no widely accepted framework for interpreting routine CGM patterns in people without diabetes, and a continuous data stream can be more confusing than clarifying (Klonoff et al., 2023; Richardson et al., 2024; Spartano et al., 2025; Yue, 2026).
Why a Meal Can Become a Glucose Wave
Postprandial glucose, meaning the glucose level after eating, is where many patterns hide. A meal containing carbohydrate breaks down into glucose that enters the bloodstream and fuels the brain, muscles, and other tissues. Insulin then escorts that glucose into cells, smoothing the curve. The system works elegantly when it works.

In insulin resistance, the muscle, liver, and fat cells respond sluggishly to insulin. The pancreas often compensates by releasing more insulin, but that compensation can erode over the years. Glucose then climbs higher after meals and lingers longer than expected, sometimes well past the next meal.

Glycemic variability describes the up-and-down movement of glucose across hours and days. Two patients can have identical average glucose levels, while one rides gentle hills and the other careens through peaks and valleys. CGM is uniquely valuable here because it shows shape, not only altitude.

The biological concern is that repeated sharp rises may strain small vessels and nerves over time. Laboratory and clinical studies link acute glucose swings to oxidative stress, which is the cellular damage that accumulates when reactive molecules overwhelm antioxidant defenses (Monnier et al., 2006; Suh & Kim, 2015). Those swings may also contribute to endothelial dysfunction, meaning impaired health of the blood-vessel lining, and to the buildup of advanced glycation end products (AGEs), which are sticky sugar-protein molecules that stiffen tissues over years.
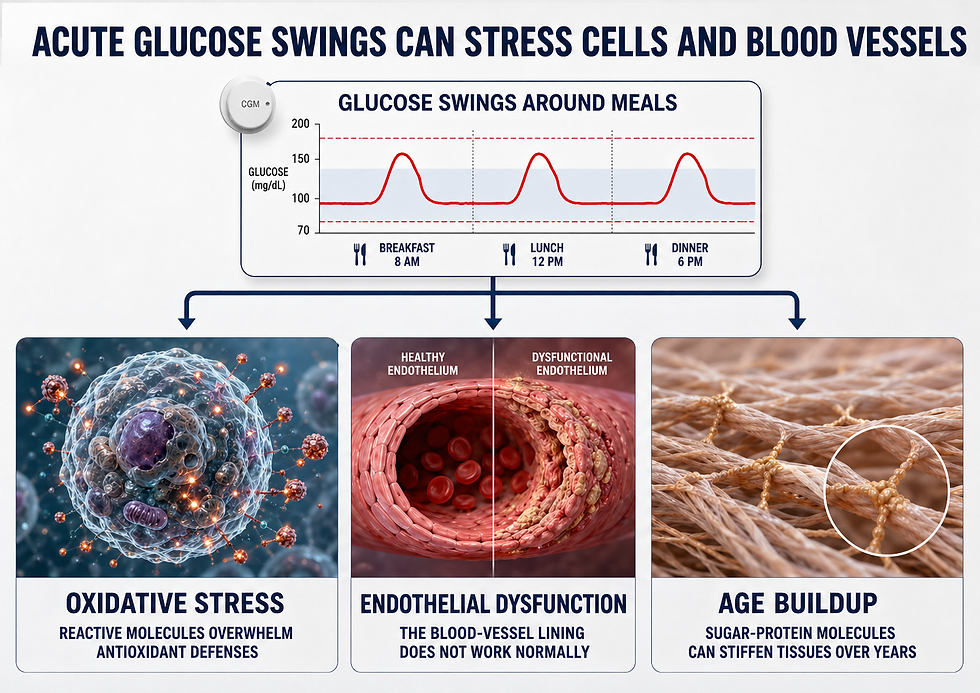
Where does 160 mg/dL fit? The International Diabetes Federation suggested a post-meal target below 160 mg/dL when it can be reached safely. Many CGM consensus reports use 70 to 180 mg/dL as the standard target range for nonpregnant adults with diabetes (Battelino et al., 2019; Ceriello & Colagiuri, 2008; International Diabetes Federation, 2011). A spike over 160 is therefore a meaningful early-warning signal, not a proven injury switch.
That nuance matters at the bedside. A clinician can reasonably ask whether repeated peaks above 160 are modifiable, especially in a patient with neuropathy risk, kidney disease, retinopathy, or cardiovascular disease. It would be too strong to claim that a single 165 mg/dL reading produces measurable damage. The clinical enemy is chronic exposure, not one imperfect dinner.
The Evidence Behind CGM in Type 2 Diabetes
The evidence for the intervention is encouraging and still maturing. In adults with Type 2 diabetes treated with basal insulin, the MOBILE randomized trial found that CGM improved A1c compared with traditional finger-stick monitoring (Martens et al., 2021). Systematic reviews and meta-analyses generally show modest A1c reduction, more time in the target range, and less time above therange, though most trials are short and not designed to capture hard complication endpoints (Jancev et al., 2024; Uhl et al., 2024).
The evidence is also expanding beyond insulin-treated diabetes. A meta-analysis of randomized trials in noninsulin-treated Type 2 diabetes documented improved glycemic control with CGM relative to usual monitoring (Ferreira et al., 2024). A more recent food-choice and self-care trial in adults not taking insulin showed substantial within-group improvements with CGM-supported self-care, though it compared two CGM-based strategies rather than CGM against no CGM (Martens et al., 2025).
CGM improves glycemic outcomes when patients receive enough education, structured feedback, and clinical follow-up to act on the data. The device alone is not the treatment. Interpretation and behavior change are the treatment pathway. Behavior-change research points in the same direction.
A systematic review of randomized trials concluded that CGM feedback can lower A1c and increase time in range, though many studies did not measure exactly which behaviors changed. Awareness appears to be the proximate mechanism, but awareness only helps when it becomes a plan (Richardson et al., 2024).
The Problem With Averages
Hemoglobin A1c (HbA1c) is a blood test that estimates average glucose exposure across roughly three months by measuring the fraction of hemoglobin that has bound glucose inside red blood cells.

It is clinically useful and historically central to diabetes care. Yet an average can hide the daily story, much as a region's average summer temperature hides every heat wave (American Diabetes Association Professional Practice Committee for Diabetes, 2026a; Centers for Disease Control and Prevention, 2024).
CGM introduces a richer vocabulary. Time in range (TIR) is the percentage of CGM readings within the target range, often 70 to 180 mg/dL for nonpregnant adults with diabetes. Time above range (TAR) is the percentage of readings that exceed the target, often 180 mg/dL. Time below range (TBR) is the percentage of time spent below target, often below 70 mg/dL, when safety becomes the dominant clinical priority (Battelino et al., 2019).

Modern CGM reports add three more useful summaries. The glucose management indicator (GMI) translates the average sensor glucose into an A1c-like estimate. The ambulatory glucose profile (AGP) compresses many days of readings into one composite daily pattern that is far easier to read than a wall of raw curves. The coefficient of variation (CV) summarizes how widely glucose values spread around the average.
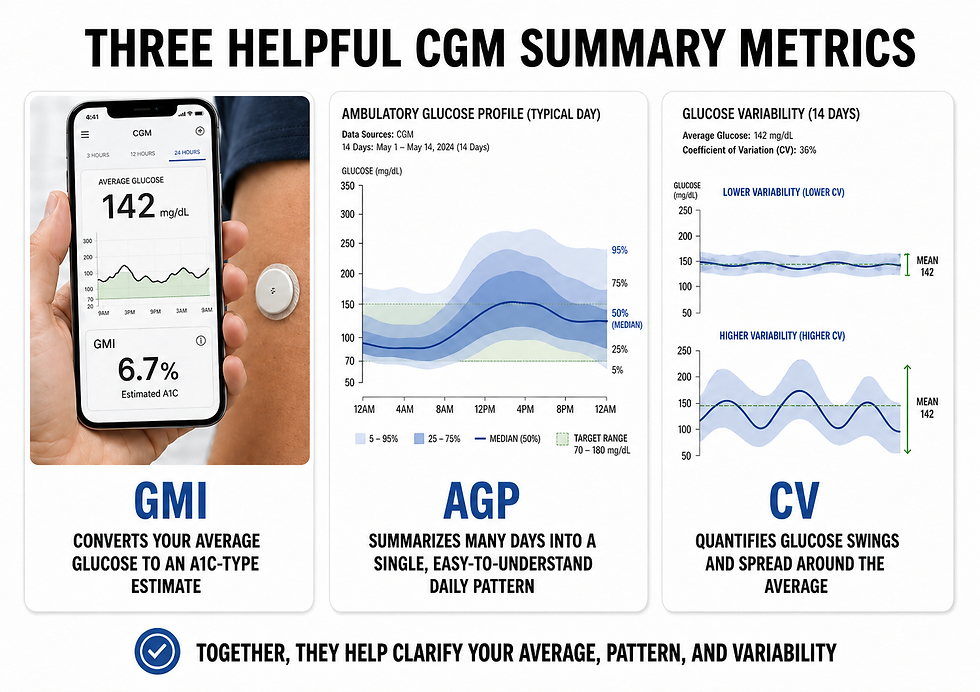
Together these measures explain a familiar paradox. A patient can post acceptable fasting readings and still experience repeated post-meal peaks between 160 and 180 mg/dL. Fasting glucose is a single morning snapshot. A1c is a three-month blur. Neither one reliably reveals whether breakfast, dinner, stress, medication timing, or late-night snacking is the source of the trouble.
The spikes are often silent. Many adults do not feel a glucose value of 165 or 175 mg/dL, especially when it lasts only thirty minutes. In a large community-based cohort, even adults without diabetes occasionally spent time above 180 mg/dL. That finding underscores why a single CGM high is not diagnostic and must be interpreted alongside meals, activity, sleep, and stress (Spartano et al., 2025).
How Spikes May Injure Small and Large Vessels
The strongest evidence for complications of CGM metrics comes from associations rather than from decades-long randomized trials. That is still clinically meaningful. A patient who spends more time above target and less time in range exposes nerves, kidneys, eyes, and vessels to more cumulative glucose stress.
Peripheral neuropathy is damage to nerves of the feet, legs, hands, or arms, often producing numbness, tingling, or burning pain.

In adults with Type 2 diabetes and chronic kidney disease, lower CGM time in range has been associated with peripheral neuropathy even when laboratory A1c was less informative (Mayeda et al., 2020). The daily pattern can matter beyond the clinic average.
Cardiovascular autonomic neuropathy (CAN) is injury to the nerves that regulate heart rate, blood pressure, and circulation.

CGM-derived time in range and time above range have both been associated with CAN in outpatients with Type 2 diabetes (Kim et al., 2021). This matters in everyday practice because autonomic symptoms often masquerade as dizziness, exercise intolerance, resting tachycardia, or orthostatic complaints.
Retinopathy refers to damage to the small blood vessels of the retina, while nephropathy refers to kidney damage that often begins with albumin leaking into the urine.

Studies in Type 2 diabetes have linked lower time in range to diabetic retinopathy and to albuminuria (Lu et al., 2018; Yoo et al., 2020). A systematic review concluded that time in range is associated with microvascular outcomes across eye, kidney, and nerve, while flagging that more prospective research is needed (Raj et al., 2022).
Macrovascular disease involves larger vessels, including coronary, cerebral, and peripheral arteries.

Causal evidence linking CGM targets directly to these outcomes is less developed. Glucose swings remain biologically plausible contributors because they appear to amplify oxidative stress, inflammation, and endothelial injury, all of which are upstream of atherosclerosis (Monnier et al., 2006; Suh & Kim, 2015).
Repeated spikes above 160 mg/dL may cumulatively increase risk, especially when part of a broader pattern of high time above range. The number invites pattern review and prevention rather than panic. One imperfect meal does not write a clinical sentence.
What CGM Adds to Behavioral Health Care
Diabetes distress is the emotional weight of living with diabetes. It includes worry, frustration, shame, burnout, and the quiet exhaustion of decisions that never end. Diabetes distress is common, prevalent across cultures and care settings, and it independently undermines glycemic control and well-being (Zu et al., 2024). Distress is also treatable.
A 2024 meta-analysis of randomized trials found that tailored psychological interventions, including cognitive behavioral therapy and mindfulness-based approaches, significantly reduced diabetes distress in adults with Type 2 diabetes, with the largest effects in the short term (Zu et al., 2024).
CGM can either ease or worsen this picture, depending on how the data are framed. The technology can reduce distress by replacing guessing with understandable feedback. It can amplify distress when it becomes a constant reminder that the body is being watched and judged. A recent ecological momentary assessment study using n-of-1 analyses found that perceived glucose levels predicted same-day diabetes distress more reliably than actual CGM-derived values (Ehrmann et al., 2024). The story patients tell themselves about their data shapes how those data feel.
For clinical psychologists and counselors, CGM functions as a behavioral map. A rise in glucose after a lonely evening snack is not just a nutritional event. It can be the visible footprint of an emotion-regulation loop that includes fatigue, deprivation, habit, reward, and self-criticism. Naming the loop is often more therapeutic than naming the carbohydrate.
Motivational interviewing is a collaborative counseling style that helps patients connect change to their own values rather than to the clinician's instruction (Miller & Rollnick, 2013). It pairs naturally with CGM because the data invite curiosity rather than compliance. Useful prompts include what the patient noticed, what surprised them, and which small adjustment feels realistic this week (Richardson et al., 2024).

Hypoglycemia, meaning low glucose, commonly defined as below 70 mg/dL, changes the safety calculus entirely. For patients using insulin or insulin-stimulating medications, the goal of reducing spikes must never become the cause of new lows. Behavioral health clinicians should encourage prompt medical coordination when CGM reveals frequent or overnight lows, or symptoms that do not match the displayed reading.
A psychologically informed CGM visit, therefore, focuses on patterns, preferences, and barriers. It avoids blame language such as "bad numbers" or "failed weeks." It also screens explicitly for perfectionism, disordered eating risk, compulsive checking, and alarm fatigue. The goal is curiosity, not surveillance.
What CGM Can Miss, Mislead, or Magnify
CGM has practical limitations that matter clinically. Because the sensor estimates glucose from interstitial fluid, rapid changes can appear late on the screen. Pressure on the sensor during sleep can sometimes produce falsely low readings, and skin irritation from adhesives is not unusual (Uhl et al., 2024).
CGM should not become a stand-alone diagnostic tool. Recent commentary has emphasized that people without diabetes lack a clear framework for interpreting every glucose fluctuation. Constant data may produce confusion and needless anxiety, with questionable public-health benefit when cost and interpretation are not addressed thoughtfully (Yue, 2026).
The same caution applies in Type 2 diabetes, although the clinical rationale for monitoring is much stronger there. A single 180 mg/dL reading should not be catastrophized. It should be placed next to the preceding meal, the medication regimen, the sleep window, the activity log, and the broader CGM report.
Not all wearables are equivalent. The FDA has explicitly warned consumers not to rely on smartwatches or smart rings that claim to measure glucose without piercing the skin, because no such devices had been authorized for that purpose at the time of the warning (U.S. Food and Drug Administration, 2024a). Clinicians should ask exactly what device the patient is using and whether it has been cleared for glucose measurement.
CGM can also amplify health inequity. Sensors incur meaningful out-of-pocket costs, insurance rules vary by state and plan, and some patients need help downloading reports or interpreting the AGP's colored bands. When ongoing access is limited, a structured professional CGM trial lasting 10 to 14 days can still be useful, particularly when the visit is built around one clear clinical question.
A Practical Workflow for Type 2 Diabetes Care
A useful workflow begins with a time-limited baseline. The patient wears CGM for 10 to 14 days while changing as little as possible about food, movement, or sleep. The goal is not perfect behavior. The goal is a clear picture of ordinary life. The next visit converts the AGP into a single clinical question. A clinician and patient might ask why breakfast produces a steep rise, why dinner stays elevated for hours, or why glucose drifts upward overnight. One focused question is more actionable than ten ambient alarms.
The first behavioral experiment should be small and safe. A patient might add protein or fiber to breakfast, reduce a portion of refined carbohydrates, or rearrange the meal. Crossover trials in adults with Type 2 diabetes show that eating vegetables and protein first, with concentrated carbohydrates consumed last, can reduce postprandial glucose peaks by roughly 50% compared with the reverse sequence (Shukla et al., 2018; Touhamy et al., 2025). The intervention is simple, requires no new food, and translates well into ordinary meals.
Movement is another high-yield experiment. Meta-analyses of post-meal exercise show that walking soon after eating blunts the glucose rise more effectively than the same walk hours later or before the meal (Engeroff et al., 2023; Kang et al., 2023; Loh et al., 2020). Even ten minutes after a meal can produce a visible flattening on the CGM trace, which makes this a perfect first experiment for a patient who needs to see results quickly. The CGM provides immediate, motivating feedback that persists in the clinician's absence.
The second experiment can address context. Sleep loss, high stress, pain flares, alcohol, infection, steroid courses, and late eating can all reshape glucose patterns. Behavioral clinicians can help patients test these variables one at a time, using the CGM as a hypothesis-checker rather than as a verdict generator.
Medication conversations belong squarely in the workflow. If TAR remains high despite feasible lifestyle changes, the CGM report serves as evidence for an informed discussion with the prescribing clinician regarding therapy adjustment. If TBR appears, especially in a patient using insulin or a sulfonylurea, a safety review should happen promptly. The CGM is not a substitute for prescribing judgment, but it is excellent shared data.
The final step is to explicitly name the success metric. For many adults with diabetes, the consensus goal is at least seventy percent TIR with minimal TBR, individualized for age, pregnancy, comorbidity, hypoglycemia risk, and treatment burden (Battelino et al., 2019). For some patients, the first meaningful win is fewer breakfast peaks, less time above 180 mg/dL, or a lower CV. Naming a small, achievable target builds the self-efficacy that sustains change.

Integrative Summary
CGM is best understood as a pattern-recognition tool. It reveals what fasting glucose and HbA1c routinely hide: the daily rhythm of meals, movement, stress, sleep, medication, and recovery. In Type 2 diabetes, that rhythm matters because repeated high exposure may, over time, contribute to nerve, vascular, kidney, and retinal injury.
The thesis is therefore largely supported, with two guardrails. First, spikes over 160 mg/dL are clinically meaningful but not a proven sharp threshold for damage. Second, CGM improves care only when the data are interpreted in the context of education, safety planning, equitable access, and psychological context.
For healthcare providers, the practical promise of CGM is not "more numbers." It is a better conversation. CGM helps patients trade vague failure narratives for testable hypotheses, smaller experiments, and more compassionate self-management. That is an outcome the technology cannot deliver on its own, but one that clinicians and patients can build together with the data it provides.
Five Takeaways
1. CGM is most powerful as patterned feedback, not as a verdict on a patient or a meal.
2. A1c and fasting glucose can look acceptable while post-meal peaks still occur between 160 and 180 mg/dL.
3. A repeated pattern above 160 mg/dL deserves clinical attention, but a single spike should not be treated as proof of injury.
4. The most useful CGM intervention is a small, safe experiment with food order, post-meal walking, sleep, stress, or medication review.
5. Behavioral health clinicians can help patients use CGM with curiosity, self-compassion, and safety rather than shame, perfectionism, or alarm fatigue.
Glossary
advanced glycation end products (AGEs): sticky molecules that form when glucose binds to proteins or fats, contributing over time to vessel and tissue stiffness.
ambulatory glucose profile (AGP): a standardized CGM report that compresses several days of glucose readings into a single, easy-to-read daily pattern.
cardiovascular autonomic neuropathy (CAN): diabetes-related injury to nerves that help regulate heart rate, blood pressure, and circulation.
coefficient of variation (CV): a percentage measure of glucose spread around the average; higher values mean greater variability.
continuous glucose monitoring (CGM): a wearable system that estimates glucose throughout the day and night using a small sensor placed under the skin.
diabetes distress: the emotional strain, frustration, worry, or burnout related to the daily burden of living with diabetes.
endothelial dysfunction: reduced ability of blood-vessel lining cells to relax vessels and regulate inflammation, clotting, and blood flow.
glucose management indicator (GMI): a CGM-derived estimate of what hemoglobin A1c might be, calculated from the average sensor glucose.
glycemic biofeedback: real-time or near-real-time glucose information used to guide behavior and treatment decisions.
glycemic variability: the degree to which glucose rises, falls, and oscillates over hours or days.
hemoglobin A1c (HbA1c): a blood test that estimates average glucose exposure over about three months by measuring the fraction of hemoglobin bound to glucose.
hypoglycemia: low glucose, commonly defined as below 70 mg/dL, which can cause shakiness, sweating, confusion, or fainting.
insulin resistance: reduced responsiveness of muscle, liver, and fat cells to insulin, making it harder to move glucose out of the bloodstream.
interstitial fluid: the watery fluid between body cells; CGM sensors estimate glucose from this fluid rather than directly from blood.
macrovascular disease: disease of larger blood vessels, including coronary, cerebral, and peripheral arteries.
motivational interviewing: a collaborative counseling style that helps patients connect health-behavior change to their own values and goals.
nephropathy: kidney damage that often begins with albumin leaking into urine and may progress to declining filtration.
oxidative stress: cellular stress that occurs when reactive molecules overwhelm antioxidant defenses and damage proteins, fats, DNA, or vessels.
peripheral neuropathy: nerve damage in the feet, legs, hands, or arms that can cause numbness, pain, tingling, or loss of protective sensation.
postprandial glucose: the glucose level after eating a meal or snack.
retinopathy: damage to small blood vessels in the retina that can impair vision.
sensor lag: the brief delay between blood glucose changes and CGM-estimated glucose in interstitial fluid.
time above range (TAR): the percentage of CGM readings above the target range, often above 180 mg/dL in diabetes care.
time below range (TBR): the percentage of CGM readings below the target range, often below 70 mg/dL.
time in range (TIR): the percentage of CGM readings within the target range, often 70 to 180 mg/dL for nonpregnant adults with diabetes.
Type 2 diabetes: a chronic metabolic condition in which insulin resistance and declining insulin production lead to elevated glucose.
References
American Diabetes Association Professional Practice Committee for Diabetes. (2026a). 6. Glycemic goals, hypoglycemia, and hyperglycemic crises: Standards of Care in Diabetes—2026. Diabetes Care, 49(Suppl. 1), S132–S149. https://doi.org/10.2337/dc26-S006 PMID: 41358894
American Diabetes Association Professional Practice Committee for Diabetes. (2026b). 7. Diabetes technology: Standards of Care in Diabetes—2026. Diabetes Care, 49(Suppl. 1), S150–S165. https://doi.org/10.2337/dc26-S007 PMID: 41358889
Battelino, T., Danne, T., Bergenstal, R. M., Amiel, S. A., Beck, R., Biester, T., Bosi, E., Buckingham, B. A., Cefalu, W. T., Close, K. L., Cobelli, C., Dassau, E., DeVries, J. H., Donaghue, K. C., Dovc, K., Doyle, F. J., III, Garg, S., Grunberger, G., Heller, S., Heinemann, L., Hirsch, I. B., Hovorka, R., Jia, W., Kordonouri, O., Kovatchev, B., Kowalski, A., Laffel, L., Levine, B., Mayorov, A., Mathieu, C., Murphy, H. R., Nimri, R., Nørgaard, K., Parkin, C. G., Renard, E., Rodbard, D., Saboo, B., Schatz, D., Stoner, K., Urakami, T., Weinzimer, S. A., & Phillip, M. (2019). Clinical targets for continuous glucose monitoring data interpretation: Recommendations from the international consensus on time in range. Diabetes Care, 42(8), 1593–1603. https://doi.org/10.2337/dci19-0028 PMID: 31177185
Centers for Disease Control and Prevention. (2024, May 15). A1C test for diabetes and prediabetes. https://www.cdc.gov/diabetes/diabetes-testing/prediabetes-a1c-test.html
Ceriello, A., & Colagiuri, S. (2008). International Diabetes Federation guideline for management of postmeal glucose: A review of recommendations. Diabetic Medicine, 25(10), 1151–1156. https://doi.org/10.1111/j.1464-5491.2008.02565.x
Ehrmann, D., Hermanns, N., Schmitt, A., Klinker, L., Haak, T., & Kulzer, B. (2024). Perceived glucose levels matter more than CGM-based data in predicting diabetes distress in type 1 or type 2 diabetes: A precision mental health approach using n-of-1 analyses. Diabetologia, 67(11), 2433–2445. https://doi.org/10.1007/s00125-024-06239-9 PMID: 39078488
Engeroff, T., Groneberg, D. A., & Wilke, J. (2023). After dinner rest a while, after supper walk a mile? A systematic review with meta-analysis on the acute postprandial glycemic response to exercise before and after meal ingestion in healthy subjects and patients with impaired glucose tolerance. Sports Medicine, 53(4), 849–869. https://doi.org/10.1007/s40279-022-01808-7 PMID: 36715875
Ferreira, R. O. M., Trevisan, T., Pasqualotto, E., Chavez, M. P., Marques, B. F., Lamounier, R. N., & van de Sande-Lee, S. (2024). Continuous glucose monitoring systems in noninsulin-treated people with type 2 diabetes: A systematic review and meta-analysis of randomized controlled trials. Diabetes Technology & Therapeutics, 26(4), 252–262. https://doi.org/10.1089/dia.2023.0390 PMID: 38090767
International Diabetes Federation. (2011). 2011 guideline for management of postmeal glucose in diabetes. International Diabetes Federation.
Jancev, M., Vissers, T. A. C. M., Visseren, F. L. J., van Bon, A. C., Serné, E. H., DeVries, J. H., de Valk, H. W., & van Sloten, T. T. (2024). Continuous glucose monitoring in adults with type 2 diabetes: A systematic review and meta-analysis. Diabetologia, 67(5), 798–810. https://doi.org/10.1007/s00125-024-06107-6 PMID: 38363342
Kang, J., Fardman, B. M., Ratamess, N. A., Faigenbaum, A. D., & Bush, J. A. (2023). Efficacy of postprandial exercise in mitigating glycemic responses in overweight individuals and individuals with obesity and type 2 diabetes—A systematic review and meta-analysis. Nutrients, 15(20), 4489. https://doi.org/10.3390/nu15204489 PMID: 37892564
Kim, M. Y., Kim, G., Park, J. Y., Choi, M. S., Jun, J. E., Lee, Y. B., Jin, S. M., Hur, K. Y., & Kim, J. H. (2021). The association between continuous glucose monitoring-derived metrics and cardiovascular autonomic neuropathy in outpatients with type 2 diabetes. Diabetes Technology & Therapeutics, 23(6), 434–442. https://doi.org/10.1089/dia.2020.0599 PMID: 33523771
Klonoff, D. C., Nguyen, K. T., Xu, N. Y., Gutierrez, A., Espinoza, J. C., & Vidmar, A. P. (2023). Use of continuous glucose monitors by people without diabetes: An idea whose time has come? Journal of Diabetes Science and Technology, 17(6), 1686–1697. https://doi.org/10.1177/19322968221110830 PMID: 35856435
Loh, R., Stamatakis, E., Folkerts, D., Allgrove, J. E., & Moir, H. J. (2020). Effects of interrupting prolonged sitting with physical activity breaks on blood glucose, insulin and triacylglycerol measures: A systematic review and meta-analysis. Sports Medicine, 50(2), 295–330. https://doi.org/10.1007/s40279-019-01183-w PMID: 31552570
Lu, J., Ma, X., Zhou, J., Zhang, L., Mo, Y., Ying, L., Lu, W., Zhu, W., Bao, Y., Vigersky, R. A., & Jia, W. (2018). Association of time in range, as assessed by continuous glucose monitoring, with diabetic retinopathy in type 2 diabetes. Diabetes Care, 41(11), 2370–2376. https://doi.org/10.2337/dc18-1131 PMID: 30201847
Martens, T., Beck, R. W., Bailey, R., Ruedy, K. J., Calhoun, P., Peters, A. L., Pop-Busui, R., Philis-Tsimikas, A., Bao, S., Umpierrez, G., Davis, G., Kruger, D., Bhargava, A., Young, L., McGill, J. B., Aleppo, G., Nguyen, Q. T., Orozco, I., Biggs, W., Lucas, K. J., Polonsky, W. H., Buse, J. B., Price, D., & Bergenstal, R. M. (2021). Effect of continuous glucose monitoring on glycemic control in patients with type 2 diabetes treated with basal insulin: A randomized clinical trial. JAMA, 325(22), 2262–2272. https://doi.org/10.1001/jama.2021.7444 PMID: 34077499
Martens, T. W., Willis, H. J., Bergenstal, R. M., Kruger, D. F., Karslioglu-French, E., & Steenkamp, D. W. (2025). A randomized controlled trial using continuous glucose monitoring to guide food choices and diabetes self-care in people with type 2 diabetes not taking insulin. Diabetes Technology & Therapeutics, 27(4), 261–270. https://doi.org/10.1089/dia.2024.0579
Mayeda, L., Katz, R., Ahmad, I., Bansal, N., Batacchi, Z., Hirsch, I. B., Robinson, N., Trence, D. L., Zelnick, L., & de Boer, I. H. (2020). Glucose time in range and peripheral neuropathy in type 2 diabetes mellitus and chronic kidney disease. BMJ Open Diabetes Research & Care, 8(1), e000991. https://doi.org/10.1136/bmjdrc-2019-000991 PMID: 31958307
Miller, W. R., & Rollnick, S. (2013). Motivational interviewing: Helping people change (3rd ed.). Guilford Press.
Monnier, L., Mas, E., Ginet, C., Michel, F., Villon, L., Cristol, J. P., & Colette, C. (2006). Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA, 295(14), 1681–1687. https://doi.org/10.1001/jama.295.14.1681 PMID: 16609090
Raj, R., Mishra, R., Jha, N., Joshi, V., Correa, R., & Kern, P. A. (2022). Time in range, as measured by continuous glucose monitor, as a predictor of microvascular complications in type 2 diabetes: A systematic review. BMJ Open Diabetes Research & Care, 10(1), e002573. https://doi.org/10.1136/bmjdrc-2021-002573 PMID: 34980591
Richardson, K. M., Jospe, M. R., Bohlen, L. C., Crawshaw, J., Saleh, A. A., &
Schembre, S. M. (2024). The efficacy of using continuous glucose monitoring as a behaviour change tool in populations with and without diabetes: A systematic review and meta-analysis of randomised controlled trials. International Journal of Behavioral Nutrition and Physical Activity, 21, 145. https://doi.org/10.1186/s12966-024-01692-6 PMID: 39716288
Shukla, A. P., Dickison, M., Coughlin, N., Karan, A., Mauer, E., Truong, W., Casper, A., Emiliano, A. B., Kumar, R. B., Saunders, K. H., Igel, L. I., & Aronne, L. J. (2018). The impact of food order on postprandial glycaemic excursions in prediabetes. Diabetes, Obesity and Metabolism, 21(2), 377–381. https://doi.org/10.1111/dom.13503 PMID: 30101510
Spartano, N. L., Sultana, N., Lin, H., Cheng, H., Lu, S., Fei, D., Murabito, J. M., Walker, M. E., Wolpert, H. A., & Steenkamp, D. W. (2025). Defining continuous glucose monitor time in range in a large, community-based cohort without diabetes. The Journal of Clinical Endocrinology & Metabolism, 110(4), 1128–1134. https://doi.org/10.1210/clinem/dgae626 PMID: 39257191
Suh, S., & Kim, J. H. (2015). Glycemic variability: How do we measure it and why is it important? Diabetes & Metabolism Journal, 39(4), 273–282. https://doi.org/10.4093/dmj.2015.39.4.273 PMID: 26301188
Touhamy, S., II, Palepu, K., Karan, A., Hootman, K. C., Riad, J., Sripadrao, S., Zhao, A. S., Giannita, A., D'Angelo, D., Alonso, L. C., Aronne, L. J., & Shukla, A. P. (2025). Carbohydrates-last food order improves time in range and reduces glycemic variability. Diabetes Care, 48(2), e15–e16. https://doi.org/10.2337/dc24-1956 PMID: 39688621
Uhl, S., Choure, A., Rouse, B., Loblack, A., & Reaven, P. (2024). Effectiveness of continuous glucose monitoring on metrics of glycemic control in type 2 diabetes mellitus: A systematic review and meta-analysis of randomized controlled trials. The Journal of Clinical Endocrinology & Metabolism, 109(4), 1119–1131. https://doi.org/10.1210/clinem/dgad652 PMID: 37987208
U.S. Food and Drug Administration. (2024a, February 21). Do not use smartwatches or smart rings to measure blood glucose levels: FDA safety communication. https://www.fda.gov/medical-devices/safety-communications/do-not-use-smartwatches-or-smart-rings-measure-blood-glucose-levels-fda-safety-communication
U.S. Food and Drug Administration. (2024b, March 5). FDA clears first over-the-counter continuous glucose monitor. https://www.fda.gov/news-events/press-announcements/fda-clears-first-over-counter-continuous-glucose-monitor
Yoo, J. H., Choi, M. S., Ahn, J., Park, S. W., Kim, Y., Hur, K. Y., Jin, S. M., Kim, G., & Kim, J. H. (2020). Association between continuous glucose monitoring-derived time in range, other core metrics, and albuminuria in type 2 diabetes. Diabetes Technology & Therapeutics, 22(10), 768–776. https://doi.org/10.1089/dia.2019.0499 PMID: 32167394
Yue, L. (2026). I study wearable health data. Here's what continuous glucose monitors miss. The Washington Post.
Zu, W., Zhang, S., Du, L., Huang, X., Nie, W., & Wang, L. (2024). The effectiveness of psychological interventions on diabetes distress and glycemic level in adults with type 2 diabetes: A systematic review and meta-analysis. BMC Psychiatry, 24(1), 660. https://doi.org/10.1186/s12888-024-06125-z PMID: 39379853

About the Author
Fred Shaffer earned his PhD in Psychology from Oklahoma State University. He earned BCIA certifications in Biofeedback and HRV Biofeedback. Fred is an Allen Fellow and Professor of Psychology at Truman State University, where he has taught for 50 years. He is a Biological Psychologist who consults and lectures in heart rate variability biofeedback, Physiological Psychology, and Psychopharmacology. Fred helped to edit Evidence-Based Practice in Biofeedback and Neurofeedback (3rd and 4th eds.) and helps to maintain BCIA's certification programs. He is a recipient of AAPB's Distinguished Scientist Award and BFE's Lifetime Impact Award.

Support Our Friends








Comments